Features and challenges of Mg
Features:
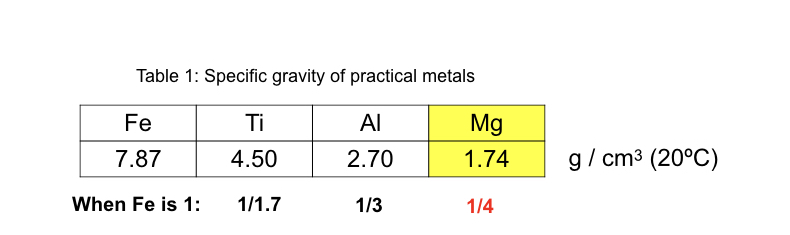
- a) The gravimetric density of Mg is about 1/4 of that of iron and about 2/3 of that of aluminum (Al). It is the lightest element among practically used structual metals. (See Table 1)
- b) This element is abundant as a resource. (Clarke number – 8, and it is the fourth among practical metals.)
- c) The human body contains Mg, making Mg high level of biocompatible material. Therefore, it can be used as medical materials for bio-implants such as bone fixators or cardiovasculars stents.
- d) Since the melting point is relatively low (at 650℃), it can easily melt and recycle.
- e) As lightweight, “beams” can be made thicker, to have higher flexural rigidity.
Challenges:
Mg alloys have been deemed disadvantageous because they have lower mechanical strength at room temperature than many aerospace grade Al alloys commonly used in aircraft, such as duralumin. Even though it is light, practical application of Mg alloys is still difficult. Low corrosion resistance (it tends to rust easily) and low ignition temperature have also been major issues.